
The alkali-metal group is group 1 or IA the alkaline-earth group is group 2 or IIA the halogen group is group 17 or VIIA and the noble-gas group is group 18 or VIIIA. But the idea of rows (periods) across which properties of elements vary and columns (groups) of elements with similar properties remains.Įach group (column) is identified by two numbers. Since Mendeleev’s time many more elements have been discovered: there are 118 in the modern periodic table below. About 150 years ago, Russian chemist Dmitri Mendeleev recognized this periodicity and created a table with horizontal rows of elements in atomic-weight order and groups of similar elements in vertical columns. Notice that the colors repeat periodically-every eight elements. The noble gases, alkali metals, alkaline earth metals, and halogens are color coded. H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Here is a list of the first twenty elements in order of increasing atomic number: (One meaning of “noble” is “unreactive”.) All are gases that undergo almost no chemical reactions, so He, Ne, Ar, Kr, and Xe are called noble gases. Just before the turn of the 20 th century, chemists discovered another group of similar elements: He, Ne, Ar, Kr, and Xe. Be, Mg, Ca, Sr, and Ba are known as alkaline earth metals or alkaline earths. Another group of elements, Be, Mg, Ca, Sr, and Ba also have similar physical properties, react with air and water (but more slowly than alkali metals), and react with halogens to form compounds with formulas like BeF 2, BeCl 2, MgCl 2, etc. ) All halogens consist of diatomic molecules, such as Cl 2. (“Halogen” comes from Greek hals, halo-”salt”. The elements F, Cl, Br, and I are called halogens. The elements Li, Na, K, Rb, and Cs are called alkali metals. (These compounds are collectively referred to as “salts” because their properties are similar to those of table salt, NaCl.) The reaction of Na(s) with Cl 2(g) to form NaCl(s) is shown in this video. In addition, all these elements react with fluorine, chlorine, bromine, and iodine to form similar compounds: LiF, NaF, KF, RbF, CsF, LiCl, NaCl, etc. I hope you have found this article helpful in solving your doubts.įeel free to ask me in the comments, if you have any doubts.As you saw in the videos previously, the elements Li, Na, K, Rb, and Cs have similar properties and react with air and water in similar ways. Then we discussed the total number of metals, Nonmetals and Metalloids present on the Periodic table. In the very beginning of this article, I showed you the Periodic table labeled with metals, Nonmetals and Metalloids. All these known elements are classified in three major categories based on their metallic and nonmetallic characteristics. Periodic table consists of total 118 elements. Get Info + HD Image of Periodic Table Summary You can effortlessly find every single detail about the elements from this single Interactive Periodic table. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Why are metalloids also known as semimetals? Free Gift for you: Interactive Periodic Table 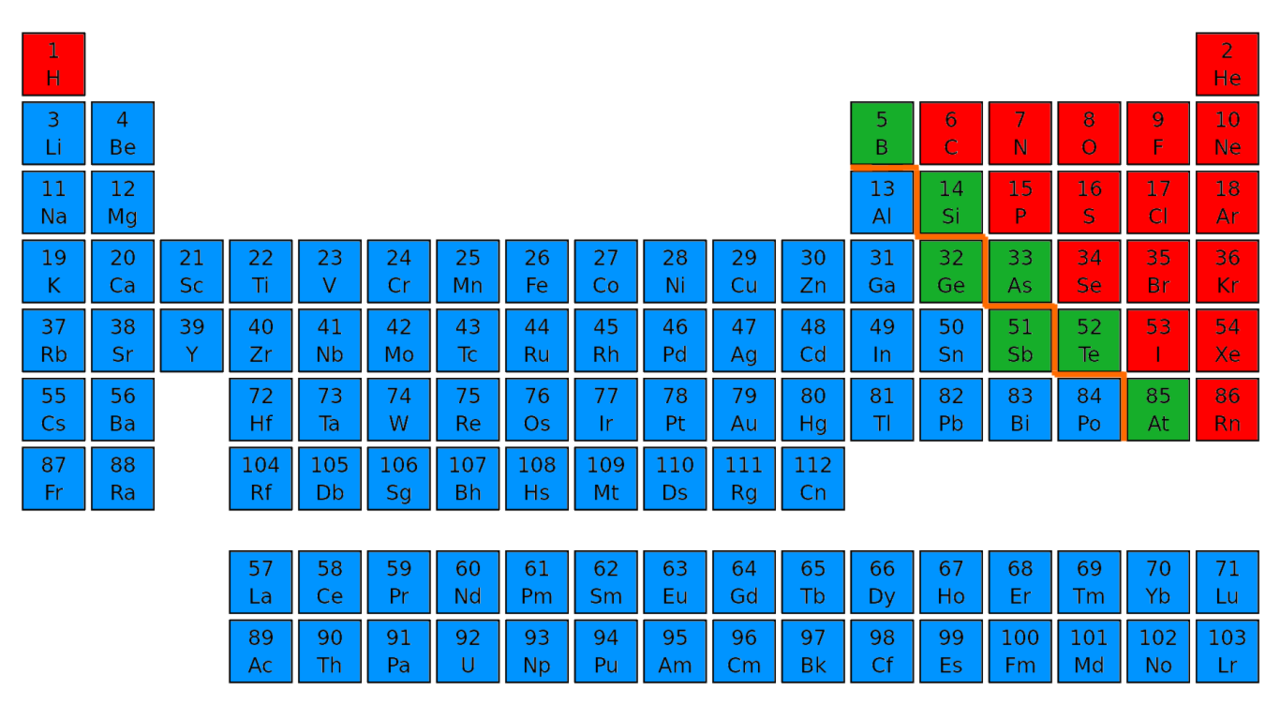
List of metalloids with atomic number, symbol and names of element.Ģ). I want to keep this article short and I have already explained the reason why astatine and polonium are not classed as metalloids. There are 6 commonly known Metalloids on the Periodic table.Īstatine and Polonium also shows some characteristics of Metalloids.īut according to some researchers, Astatine and Polonium are not classes as Metalloids. How many Metalloids are on the Periodic table? They are found between the metals and nonmetals.Įlements which have the metallic properties as well as nonmetallic properties are classified as Metalloids.īecause of this reason, they are also known as semimetals. Above picture shows you the labeled Metalloids on the Periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
